Metabolomics is transforming our understanding of biology by profiling small molecules that reflect physiological and pathological states. As the field matures, consistent and transparent practices become essential to turning complex data into reliable scientific knowledge. Metabolomics standards are central to this effort: they ensure data quality, make experiments comparable across labs, and accelerate discovery by enabling data reuse. In this post, we explain why these standards matter, what they cover, and how organizations like IROA Technologies contribute to building a more reproducible metabolomics ecosystem.
What are metabolomics standards?
Metabolomics standards are agreed-upon guidelines, formats, and best practices for designing experiments, collecting and processing data, and reporting results. They include:
- Standardized sample collection and storage protocols.
- Instrument calibration and quality-control procedures.
- Data formats and metadata requirements to describe experimental context.
- Statistical and bioinformatic processing pipelines with versioned software and parameters.
- Reporting guidelines that specify what must be shared for interpretation and reuse.
Together, these elements reduce variability that comes from pre-analytical handling, instrument differences, and divergent data processing choices.
Why reproducibility is a pressing issue
Reproducibility—the ability of independent researchers to obtain the same findings using the same methods—is a cornerstone of science. However, metabolomics faces several challenges:
- Biological complexity: Metabolites are influenced by diet, circadian rhythms, microbiome composition, and environmental factors.
- Technical variability: Different mass spectrometers, NMR instruments, chromatographic conditions, and vendor software produce data with varying characteristics.
- Data processing diversity: Multiple peak-picking, normalization, and identification approaches can yield different outputs from the same raw data.
- Incomplete reporting: Without metadata and methodological detail, reproducing or reanalyzing results is difficult or impossible.
Adopting metabolomics standards addresses these sources of variability by making experimental context explicit and by harmonizing critical steps across studies.
Practical benefits of standards
- Improved data quality: Standard operating procedures for sample handling and instrument QC reduce noise and systematic bias.
- Easier data sharing and reuse: Standard data formats and metadata enable databases and repositories to accept, index, and make data discoverable.
- Faster validation and translation: Clear reporting and reproducible pipelines speed up independent validation, meta-analyses, and clinical translation.
- Regulatory and clinical readiness: For biomarker development, regulators expect traceability, documented QC, and reproducible methods—standards support these requirements.
- Cost and time savings: Preventing avoidable variation reduces the need for repeated experiments and troubleshooting.
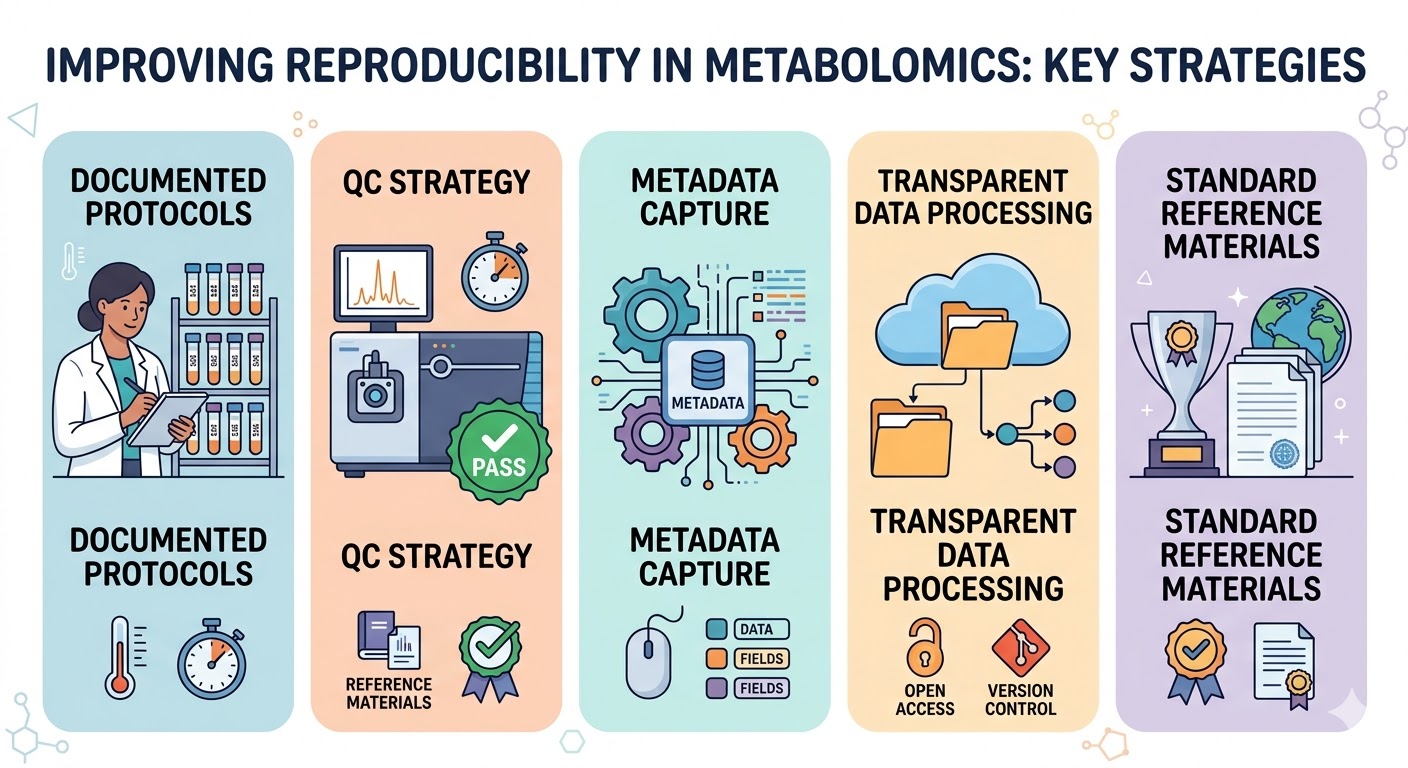
Key components to implement in your lab
- Documented protocols: Keep up-to-date SOPs for sample collection, storage, and extraction. Include details such as anticoagulants, time-to-freezing, and freeze–thaw limits.
- QC strategy: Use pooled quality-control samples, internal standards, and blanks. Monitor instrument performance over time and set acceptance criteria.
- Metadata capture: Record participant demographics, collection conditions, instrument settings, and software versions. Machine-readable metadata (e.g., ISA-Tab or mzML annotations) improve downstream interoperability.
- Transparent data processing: Share raw data, processing scripts, and parameter settings. Containerization or workflow managers (e.g., Nextflow, Snakemake) help preserve reproducible pipelines.
- Standard reference materials: Use certified reference materials or well-characterized mixtures for system suitability testing and inter-lab comparisons.
Role of industry and providers
Companies and technology providers play a pivotal role in disseminating and operationalizing standards. For example, IROA Technologies develops tools and products that help labs implement robust QA/QC workflows and standardized data capture. By integrating reference materials, software that enforces metadata fields, and reporting templates, industry partners can lower the barrier for labs to adopt best practices. Collaboration between academia, industry, and standards bodies is crucial to create practical, widely accepted guidelines.
Community initiatives and resources
Several community-driven efforts and repositories support metabolomics standards and FAIR data principles (Findable, Accessible, Interoperable, Reusable). Examples include the Metabolomics Standards Initiative (MSI) and public repositories that accept standardized submissions. Engaging with these groups and contributing to consensus documents helps align methods across the field. For up-to-date reporting standards and submission guidelines, consult resources such as the MetaboLights repository .
Overcoming common barriers
Adoption of standards can be slowed by perceived cost, time investment, and legacy workflows. Practical strategies to overcome these barriers include:
- Start small: Implement key QC checks and metadata capture first, then expand protocols.
- Leverage automation: Use laboratory information management systems (LIMS) or vendor tools to capture metadata automatically.
- Train personnel: Regular training ensures protocols are followed consistently.
- Use community templates: Adopt existing reporting templates rather than building from scratch.
Final thoughts
Metabolomics standards are not bureaucratic red tape—they are practical tools that strengthen the credibility, impact, and translational potential of metabolomics research. By reducing technical noise, enabling data reuse, and facilitating collaboration, standards transform isolated experiments into reliable knowledge. Whether you are a bench scientist, bioinformatician, or technology provider such as IROA Technologies, investing in standards pays dividends in reproducibility and scientific progress.
FAQs
Q: What is the single most important step to improve reproducibility in metabolomics?
A: Consistently applying quality-control samples and documenting instrument and sample handling metadata are among the highest-impact steps—together they reveal and reduce technical variability.
Q: How often should QC samples be run?
A: A common approach is to inject pooled QC samples at regular intervals (for example, every 5–10 study samples) to monitor instrument drift; the exact frequency depends on run length and instrument stability.
Q: Are there standard file formats I should use for metabolomics data?
A: Yes—open formats such as mzML for mass spectrometry and nmrML for NMR help ensure interoperability and long-term accessibility.
Q: Can small labs adopt these standards without large budgets?
A: Absolutely. Many standards are procedural (documentation, metadata capture) and require minimal cost. Affordable reference materials and free community tools can also support adoption.
Q: How do companies like IROA Technologies help with standards?
A: Providers can supply reference materials, QC products, software for metadata capture, and consulting to implement validated workflows—reducing the technical burden on individual labs.