Modern metabolomics relies heavily on precision—both in identifying molecules and accurately quantifying them within complex biological samples. One of the most advanced approaches enabling this level of accuracy is Isotopic Ratio Outlier Analysis (). At the heart of this methodology lies a powerful concept known as isotopic envelopes, which significantly improves both identification and quantitation in mass spectrometry-based studies.
In this blog, we’ll explore how works, the science behind isotopic labeling, and how isotopic envelopes contribute to enhanced precision in metabolomics,
Understanding: A Smarter Approach to Metabolomics
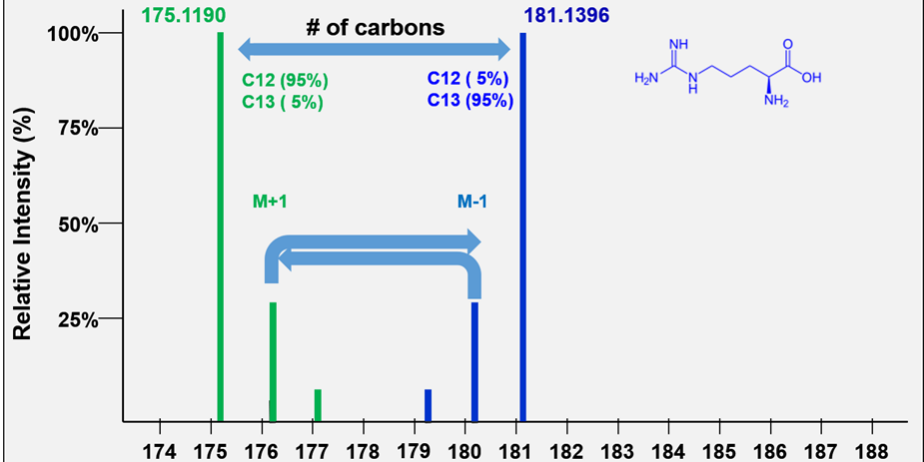
is designed to create distinct molecular signatures within a biological sample. This is achieved by incorporating stable carbon isotopes—¹²C and ¹³C—into molecules at controlled ratios. Typically, one population of molecules is labeled with approximately 5% ¹³C, while another is labeled with around 95% ¹³C.
This dual-labeling strategy produces a mirror-image pattern in mass spectrometry. Molecules with 5% ¹³C show an enhanced M+1 peak, while those with 95% ¹³C display an enhanced M-1 peak. These mirrored patterns are not just visually distinctive—they are analytically powerful.
Compared to traditional labeling methods (which often exceed 98% enrichment), enhances minor peaks that would otherwise be difficult to detect. For example, in a six-carbon molecule like arginine, the M+1 or M-1 peaks can reach up to 32% of the monoisotopic peak height, compared to just 6% under natural abundance conditions. This increased visibility plays a crucial role in accurate data interpretation.
Isotopomers and Isotopologs: Building the Foundation
To understand isotopic envelopes, it’s important to distinguish between isotopomers and isotopologs:
- Isotopomers are molecules with the same number of isotopes but arranged differently. They share the same mass and appear as a single peak in mass spectra.
- Isotopologs, on the other hand, differ in the number of isotopes they contain. Each isotopolog has a unique mass and appears as a separate peak.
Every molecule forms an isotopolog “ladder” in a mass spectrum, starting from the ¹²C monoisotopic peak and extending to the ¹³C monoisotopic peak. The number of steps in this ladder depends on the number of carbon atoms in the molecule.
This ladder is unique to a molecular formula, making it a reliable tool for identification. However, interpreting these patterns can become complex—this is where isotopic envelopes simplify the process.
What Are Isotopic Envelopes?
In simple terms, isotopic envelopes are the grouped patterns of peaks formed by isotopologs in a mass spectrum. Instead of analyzing individual peaks, scientists consider the entire cluster of peaks as a single analytical unit.
Each molecule generates a characteristic isotopic envelope based on its carbon composition and isotopic distribution. In experiments, two envelopes are always present:
- A natural or lightly labeled sample (e.g., 5% ¹³C)
- A heavily labeled internal standard (e.g., 95% ¹³C)
These two envelopes overlap on the same isotopolog ladder but differ in their peak distributions, creating a distinctive and recognizable pattern.
Enhancing Identification Through Unique Patterns
One of the key advantages of isotopic envelopes is their ability to act as molecular fingerprints. Since each molecular formula produces a unique isotopolog ladder and envelope shape, researchers can confidently identify compounds even in complex mixtures.
Additionally, the mass difference between peaks—approximately 1.00335 atomic mass units—corresponds to the difference between ¹²C and ¹³C. This consistent spacing helps analytical tools, such as ClusterFinder developed by technologies, automatically detect and classify these patterns.
This level of precision reduces ambiguity and minimizes false identifications, which is critical in metabolomics research.
 Improving Quantitation Accuracy
Improving Quantitation Accuracy
Accurate quantitation is just as important as identification. Traditional methods often rely on the intensity of a single peak, which can be misleading due to signal suppression or noise.
takes a different approach. Instead of focusing on one peak, it sums all peaks within each isotopic envelope. This provides a more reliable measure of the total molecule concentration.
An important principle here is that the ratio between the ¹²C and ¹³C envelopes remains consistent, even if signal suppression occurs. This allows for built-in correction mechanisms, ensuring that quantitation remains accurate across different samples.
Furthermore, advanced algorithms normalize the data to account for variability between experiments, delivering highly reproducible results.
The Role of Mass Defect in Precision
Another factor that enhances the accuracy of -based analysis is the concept of mass defect. This refers to the small difference between the nominal mass and the exact mass of a molecule.
By analyzing both the ¹²C and ¹³C monoisotopic peaks, researchers can calculate the residual mass contributed by other elements in the molecule. Averaging these values reduces error and helps determine the exact molecular formula.
With high-resolution mass spectrometry, this approach often leads to a single possible formula for molecules under 500 amu, further improving confidence in the results.
For a deeper understanding of mass spectrometry and isotopic analysis, you can explore this resource:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2928650/
Why This Matters in Real-World Applications
The ability to accurately identify and quantify metabolites has far-reaching implications. From disease research and drug development to environmental analysis and nutrition studies, precision is essential.
By leveraging isotopic envelopes, provides:
- Greater sensitivity in detecting low-abundance compounds
- Improved accuracy in quantitation
- Reduced analytical errors
- Enhanced reproducibility across experiments
These benefits make it a valuable tool for researchers and laboratories aiming for high-quality data.
Conclusion
represents a significant advancement in metabolomics, offering a robust framework for both identification and quantitation. At the core of this methodology, isotopic envelopes simplify complex spectral data and provide a reliable means of interpreting molecular information.
By combining controlled isotopic labeling, unique peak patterns, and advanced computational tools, iroa technologies continue to push the boundaries of analytical precision. As metabolomics continues to evolve, approaches like will play an increasingly important role in delivering accurate and actionable insights.
Frequently Asked Questions (FAQs)
- What are isotopic envelopes in metabolomics?
Isotopic envelopes are clusters of mass spectral peaks formed by molecules with different isotopic compositions. They help in identifying and quantifying compounds more accurately. - How does IROA improve quantitation accuracy?
IROA improves accuracy by summing all peaks within an isotopic envelope rather than relying on a single peak, reducing errors caused by signal suppression. - Why are 5% and 95% ¹³C labels used in IROA?
These specific ratios create distinct mirror-image patterns in mass spectra, making it easier to identify and differentiate molecules. - What is the difference between isotopomers and isotopologs?
Isotopomers have the same number of isotopes but different arrangements, while isotopologs differ in the number of isotopes and have different masses. - How do isotopic envelopes help in compound identification?
They provide unique and predictable patterns for each molecular formula, allowing researchers to accurately identify compounds even in complex mixtures.

 Improving Quantitation Accuracy
Improving Quantitation Accuracy



