In the world of modern science, identifying molecules with precision is essential for breakthroughs in healthcare, environmental studies, and biotechnology. One of the most powerful tools scientists rely on for this purpose is mass spectrometry. While this technique may sound complex, one of its most fascinating aspects—the isotopic envelope—plays a crucial role in making molecular identification highly accurate.
In this blog, we’ll break down this concept in simple terms and explore how it helps scientists achieve reliable results in analytical chemistry.
What Is an Isotopic Envelope?
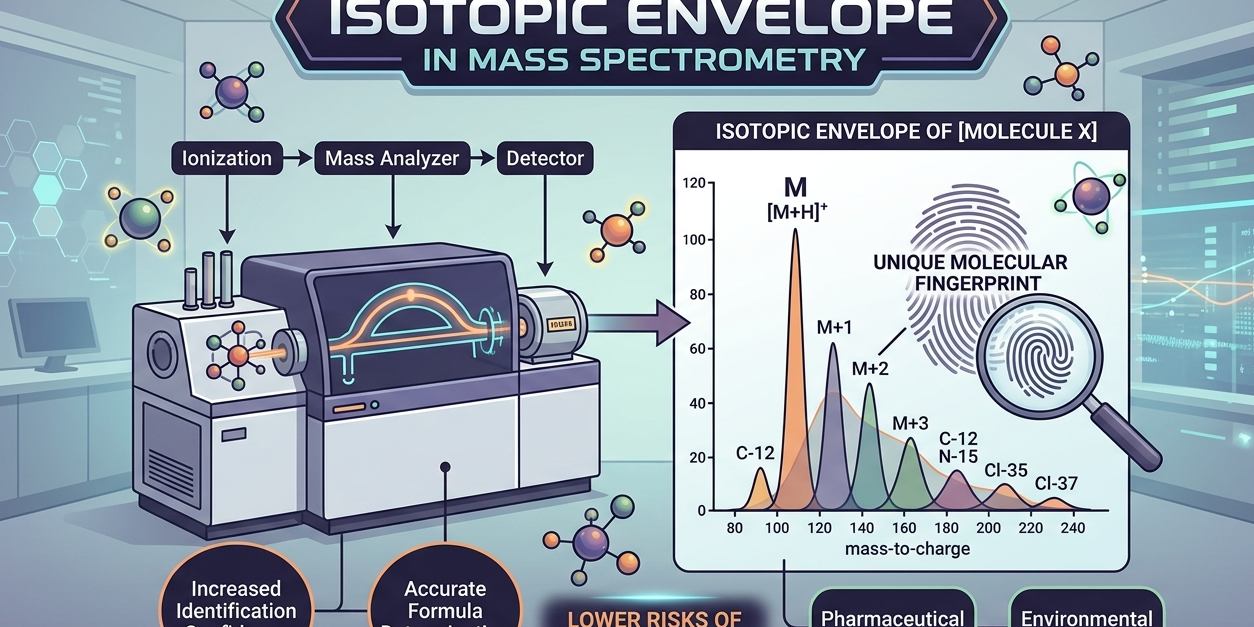
Every chemical element exists in different forms known as isotopes. These isotopes have the same number of protons but different numbers of neutrons, which means they have slightly different masses. When a molecule is analyzed using mass spectrometry, it doesn’t appear as a single peak. Instead, it produces a cluster of peaks representing its isotopic variations. This cluster is called the isotopic envelope.
Think of it like a fingerprint. Just as every person has a unique fingerprint, every molecule has a distinct pattern formed by its isotopes. This pattern helps scientists confirm the identity of a molecule with remarkable accuracy.
Why Accurate Molecular Identification Matters
Accurate molecular identification is critical across multiple industries. In pharmaceuticals, it ensures that drugs are pure and safe. In environmental science, it helps detect pollutants at very low concentrations. In metabolomics, it allows researchers to understand complex biological processes.
Without precise identification methods, even small errors can lead to incorrect conclusions. That’s where the isotopic envelope becomes incredibly valuable—it adds an extra layer of verification.
How the Isotopic Envelope Works in Mass Spectrometry
Mass spectrometry measures the mass-to-charge ratio of ions. When a sample is introduced into the instrument, molecules are ionized and broken into charged particles. These particles are then detected and displayed as peaks on a graph.
Instead of showing just one peak, a molecule produces multiple peaks due to the presence of isotopes like carbon-13 or nitrogen-15. The spacing and intensity of these peaks form a predictable pattern. Scientists analyze this pattern to confirm the molecular structure.
For example, a molecule containing many carbon atoms will show a noticeable contribution from carbon-13 isotopes. By examining the pattern, researchers can estimate the number of atoms and verify whether the detected compound matches the expected one.
 Enhancing Confidence in Results
Enhancing Confidence in Results
One of the biggest advantages of using isotopic patterns is the increased confidence in analytical results. When scientists rely only on a single mass value, there is always a chance of overlap with another compound of similar mass. However, when the full isotopic pattern is considered, the likelihood of misidentification drops significantly.
This is especially important in complex samples, such as biological fluids or environmental extracts, where thousands of compounds may be present. The isotopic envelope helps distinguish between molecules that might otherwise appear identical.
Applications Across Industries
The use of isotopic patterns extends across many scientific fields:
- Pharmaceutical Research: Ensures drug compounds are correctly identified and free from contaminants.
- Metabolomics: Helps researchers study metabolic pathways and understand disease mechanisms.
- Environmental Testing: Detects trace-level pollutants with high accuracy.
- Food Safety: Identifies adulterants and ensures product authenticity.
Companies like iroa technologies are at the forefront of advancing analytical techniques that leverage isotopic data to deliver precise and reproducible results. Their innovations help scientists gain deeper insights into complex datasets.
The Role of Advanced Technologies
Modern advancements in mass spectrometry have made it easier to analyze isotopic patterns with greater resolution and sensitivity. High-resolution instruments can distinguish even the smallest differences in mass, making the isotopic envelope more detailed and informative.
Additionally, software tools now automate the interpretation of these patterns, saving time and reducing human error. This combination of hardware and software advancements has significantly improved the reliability of molecular identification.
For a deeper understanding of mass spectrometry and its applications, you can explore this helpful resource:
https://www.britannica.com/science/mass-spectrometry
Challenges and Considerations
While isotopic analysis is powerful, it does come with challenges. Interpreting complex patterns requires expertise and high-quality instrumentation. In some cases, overlapping signals from different molecules can complicate the analysis.
However, ongoing research and technological improvements continue to address these challenges. With better algorithms and more advanced instruments, scientists are now able to extract meaningful insights even from highly complex samples.
Why It Matters for the Future
As scientific research becomes more data-driven, the need for accurate and reliable analytical methods will only grow. The isotopic envelope will continue to play a key role in ensuring that molecular identification remains precise and trustworthy.
From developing new medicines to monitoring environmental health, this concept supports a wide range of applications that impact our daily lives. Its importance will only increase as new discoveries push the boundaries of science.
FAQs
1. What is the purpose of an isotopic envelope in mass spectrometry?
It helps identify molecules by providing a unique pattern of isotopic peaks, improving accuracy and confidence in results.
2. Why don’t molecules appear as a single peak?
Because elements have isotopes with different masses, molecules produce multiple peaks instead of just one.
3. How does the isotopic envelope improve accuracy?
It allows scientists to confirm a molecule’s identity by matching both its mass and its isotopic pattern.
4. Where is this concept commonly used?
It is widely used in pharmaceuticals, environmental testing, metabolomics, and food safety analysis.
5. Do all molecules have an isotopic pattern?
Yes, all molecules made of elements with isotopes will produce a pattern, though its complexity varies depending on the composition.

 Enhancing Confidence in Results
Enhancing Confidence in Results