Drug discovery has evolved dramatically over the past few decades, driven by advances in biotechnology, computational tools, and molecular biology. Among the many innovations shaping this field, peptide-based research has emerged as a powerful approach for identifying new therapeutic candidates. At the heart of this progress lies a critical tool: Standard Peptide Libraries. These curated collections of peptides are helping scientists accelerate research, improve accuracy, and uncover new possibilities in medicine.
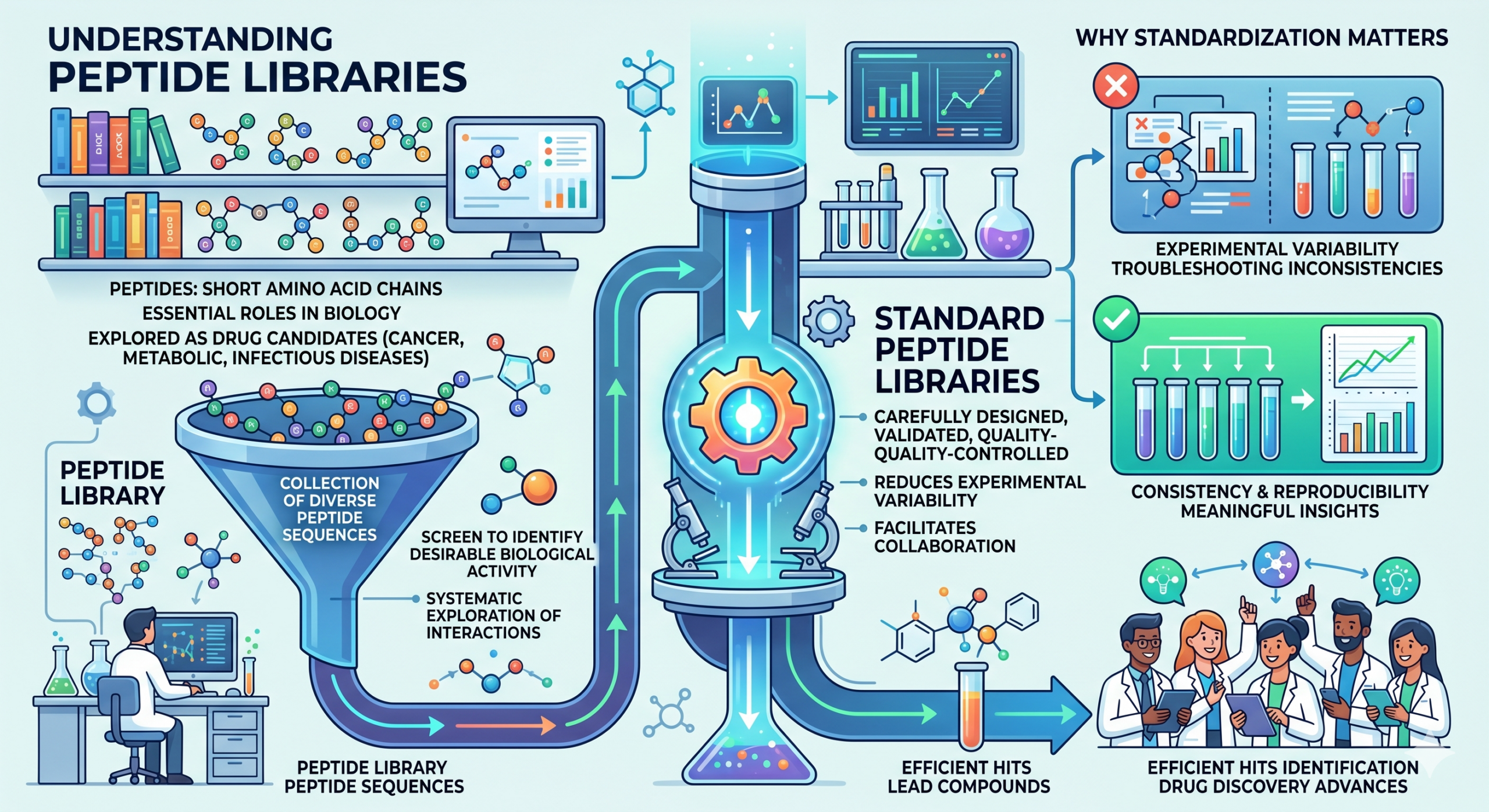
Understanding Peptide Libraries
Peptides are short chains of amino acids that play essential roles in biological processes. Because of their specificity and versatility, peptides are increasingly being explored as drug candidates for a wide range of diseases, including cancer, metabolic disorders, and infectious diseases.
A peptide library is essentially a collection of diverse peptide sequences that researchers can screen to identify those with desirable biological activity. These libraries can vary in size, complexity, and design, but they all serve a common purpose: enabling systematic exploration of peptide interactions with biological targets.
Why Standardization Matters
In drug discovery, consistency and reproducibility are crucial. This is where Standard Peptide Libraries stand out. Unlike custom or random peptide collections, standardized libraries are carefully designed, validated, and quality-controlled. This ensures that researchers across different labs and studies can rely on consistent data and comparable results.
Standardization reduces experimental variability, allowing scientists to focus on meaningful insights rather than troubleshooting inconsistencies. It also facilitates collaboration, as teams can work with the same reference materials and share findings more effectively.
Accelerating Target Identification
One of the most important steps in drug discovery is identifying molecular targets—specific proteins or receptors associated with a disease. Peptide libraries enable high-throughput screening against these targets, helping researchers quickly identify peptides that bind with high affinity.
This process significantly shortens the time required to move from hypothesis to discovery. Instead of testing individual compounds one by one, scientists can screen thousands of peptides simultaneously. This efficiency is particularly valuable in early-stage research, where speed and accuracy can make a significant difference.
Enhancing Drug Design and Optimization
Once a promising peptide is identified, it often needs to be optimized to improve its stability, potency, and bioavailability. Standardized libraries provide a structured framework for this optimization process. Researchers can compare similar peptide sequences, analyze structure-activity relationships, and make informed modifications.
This iterative approach leads to more effective drug candidates with fewer side effects. It also reduces the risk of failure in later stages of development, saving both time and resources.
Supporting Proteomics and Biomarker Discovery
Beyond drug development, peptide libraries play a vital role in proteomics—the large-scale study of proteins. By using standardized peptide sets, scientists can improve the accuracy of protein identification and quantification.
This is particularly important for biomarker discovery, where researchers aim to identify molecular signatures associated with specific diseases. Reliable peptide references ensure that findings are reproducible and clinically relevant, paving the way for better diagnostics and personalized medicine.
Integration with Advanced Technologies
Modern drug discovery relies heavily on advanced technologies such as mass spectrometry, machine learning, and high-throughput screening platforms. Standard Peptide Libraries integrate seamlessly with these tools, enhancing their effectiveness.
For example, in mass spectrometry-based proteomics, standardized peptides serve as calibration and validation tools. This improves data quality and enables more precise measurements. Similarly, machine learning algorithms can use standardized datasets to identify patterns and predict peptide behavior, further accelerating research.
Organizations like iroa technologies are contributing to this space by developing innovative solutions that combine metabolomics and proteomics with standardized approaches. Their work highlights the growing importance of reliable, high-quality tools in advancing scientific discovery.
Reducing Costs and Increasing Efficiency
Drug discovery is an expensive and time-consuming process, often taking years and billions of dollars to bring a new drug to market. By improving efficiency and reducing experimental errors, standardized peptide libraries help lower these costs.
Researchers can avoid redundant experiments, minimize resource wastage, and streamline workflows. This not only benefits pharmaceutical companies but also accelerates the delivery of new treatments to patients in need.
Real-World Applications
The impact of peptide libraries can be seen across various therapeutic areas. In oncology, they are used to identify peptides that target cancer cells with high specificity. In infectious diseases, they help discover antimicrobial peptides that can combat resistant pathogens.
Additionally, peptide-based vaccines are gaining attention, especially in the wake of global health challenges. Standardized libraries enable rapid screening and development of vaccine candidates, making them a valuable tool in public health efforts.
For further reading on peptide-based drug discovery, you can explore this informative resource:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6078856/
Challenges and Future Outlook
Despite their advantages, peptide libraries are not without challenges. Designing comprehensive libraries that cover all possible sequences can be complex. Additionally, peptides may face issues such as instability and rapid degradation in the body.
However, ongoing research is addressing these limitations. Advances in peptide engineering, delivery systems, and computational modeling are expanding the potential of peptide-based therapies. As these technologies continue to evolve, the role of standardized libraries will only become more significant.
Conclusion
In the rapidly advancing field of drug discovery, tools that enhance efficiency, accuracy, and collaboration are invaluable. Standard Peptide Libraries provide a robust foundation for exploring peptide-based therapeutics, enabling researchers to identify, optimize, and validate drug candidates more effectively.
By integrating with modern technologies and supporting a wide range of applications, these libraries are transforming how scientists approach complex biological challenges. With continued innovation from companies like iroa technologies, the future of drug discovery looks increasingly promising.
FAQs
1. What are peptide libraries used for?
Peptide libraries are used to screen and identify peptides that interact with specific biological targets, aiding in drug discovery, diagnostics, and research.
2. Why are standardized peptide libraries important?
They ensure consistency, reproducibility, and reliability in experiments, making research findings more accurate and comparable across studies.
3. How do peptide libraries help in drug discovery?
They enable high-throughput screening, accelerate target identification, and support optimization of drug candidates.
4. Are peptide-based drugs widely used today?
Yes, peptide-based drugs are increasingly used in treating conditions like diabetes, cancer, and hormonal disorders.
5. What challenges do peptide therapies face?
Common challenges include stability, delivery, and potential degradation in the body, though ongoing research is addressing these issues.