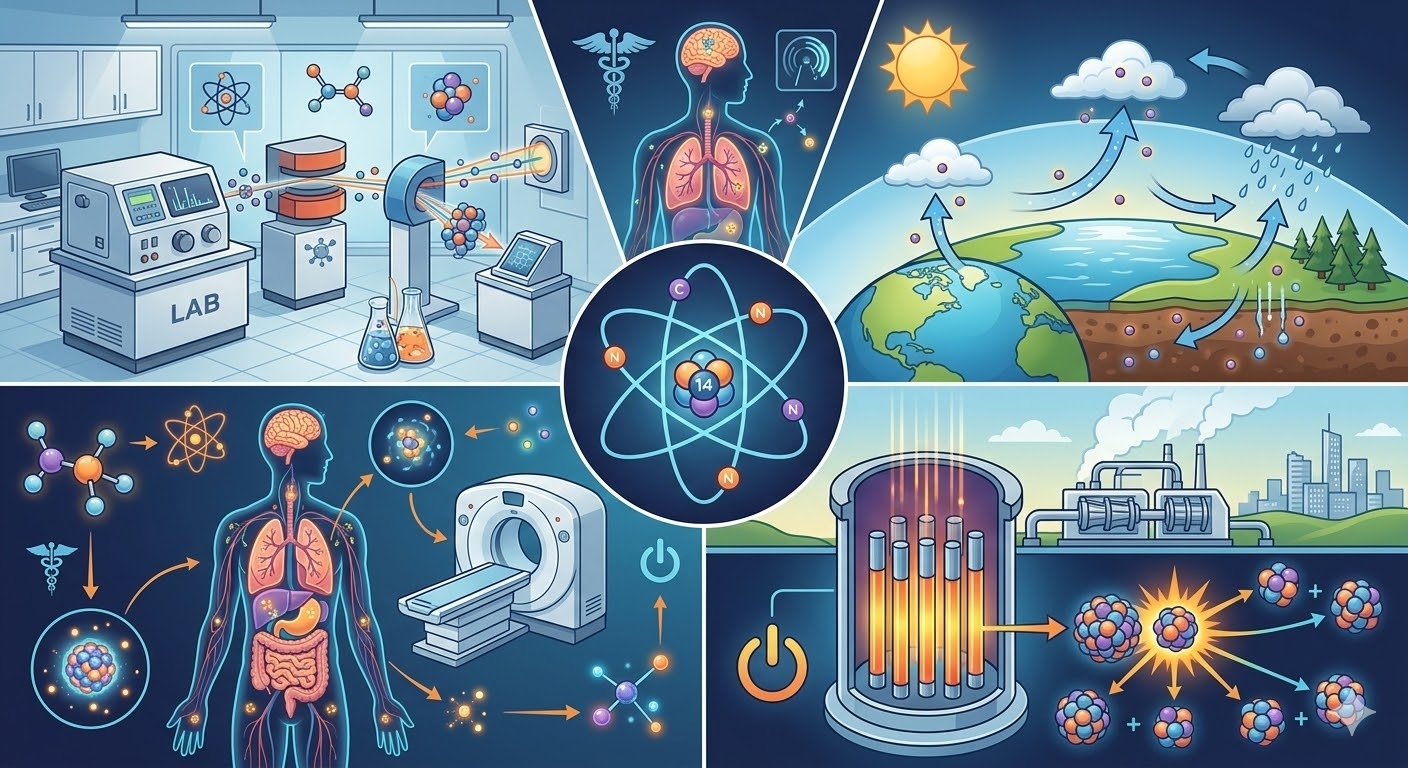
Nuclear chemistry is a fascinating field that focuses on the structure and behavior of atomic nuclei. It helps scientists understand radioactive elements, nuclear reactions, and how atoms transform during nuclear processes. One concept that plays a crucial role in this field is Isobaric Compounds. These atomic species share the same mass number but differ in their atomic numbers, which means they contain different numbers of protons and neutrons.
Understanding this concept is essential for scientists working in nuclear science, medicine, environmental studies, and analytical chemistry. By studying these atomic forms, researchers can track nuclear reactions, identify radioactive materials, and even develop medical treatments. In this article, we will explore why these compounds are so important in nuclear chemistry and how they are used in real-world applications.
Understanding the Concept in Nuclear Chemistry
In nuclear chemistry, atoms are often classified based on their number of protons, neutrons, and mass numbers. When atoms have the same total mass number but different atomic numbers, they fall into the category of Isobaric Compounds.
For example, carbon-14 and nitrogen-14 share the same mass number of 14 but contain different numbers of protons and neutrons. Because of this, they behave differently chemically while still having the same atomic mass.
This unique property makes them extremely valuable in nuclear research. Scientists can use them to trace nuclear transformations and analyze the stability of atomic nuclei.
Role in Understanding Nuclear Reactions
One of the main reasons these compounds are important in nuclear chemistry is their role in studying nuclear reactions. During radioactive decay or nuclear transformations, one element can change into another while maintaining the same mass number.
For instance, beta decay often produces atoms with identical mass numbers but different atomic numbers. By analyzing such changes, scientists can better understand how nuclear reactions occur and how radioactive materials behave.
These studies help researchers predict nuclear stability, identify reaction pathways, and improve nuclear energy systems. As a result, the study of Isobaric Compounds contributes significantly to advancing nuclear science.
Importance in Radiometric Dating
Another important application of these compounds is in radiometric dating. This technique is widely used in geology, archaeology, and environmental science to determine the age of materials.
Some radioactive atoms decay into other elements that have the same mass number. By measuring the ratio between parent and daughter atoms, scientists can estimate the age of fossils, rocks, or ancient artifacts.
Radiometric dating techniques such as carbon dating rely on these nuclear transformations to provide accurate age estimates. For more details on nuclear dating methods, you can explore this resource from the International Atomic Energy Agency:
https://www.iaea.org
Understanding these transformations would not be possible without studying atomic forms with identical mass numbers.
Applications in Nuclear Medicine
Modern medicine relies heavily on nuclear chemistry, especially in diagnostic imaging and cancer treatment. Certain radioactive atoms used in medical imaging can transform into atoms with the same mass number but different atomic structures.
These nuclear transformations allow doctors to trace radioactive materials in the body and monitor organ function. For example, radioactive tracers used in PET scans or other imaging technologies depend on predictable nuclear behavior.
Research on Isobaric Compounds helps scientists design better diagnostic tools and safer medical isotopes. As a result, patients benefit from more accurate diagnoses and improved treatment methods.
Use in Environmental and Atmospheric Studies
Nuclear chemistry is also used to monitor environmental changes and pollution. Certain radioactive atoms naturally occur in the atmosphere and the Earth’s crust. When these atoms undergo nuclear transformations, they can form atoms with the same mass number.
By studying these changes, scientists can track environmental processes such as atmospheric circulation, soil formation, and water movement.
Environmental scientists use nuclear data to analyze pollution sources, climate patterns, and ecological changes. This makes nuclear chemistry a valuable tool for understanding the Earth’s natural systems.
Role in Advanced Analytical Technologies
Modern laboratories rely on advanced analytical tools to study atomic structures and nuclear behavior. Technologies such as mass spectrometry allow scientists to identify atoms with identical mass numbers but different atomic compositions.
Companies like Iroa technologies contribute to scientific research by developing advanced analytical solutions that help researchers analyze complex chemical data. These technologies support scientists in identifying subtle nuclear differences and understanding atomic transformations.
Such innovations are especially useful in research fields like nuclear chemistry, pharmaceutical development, and environmental monitoring.
Contribution to Nuclear Energy Research
Nuclear energy remains one of the most powerful sources of electricity in the modern world. However, developing safe and efficient nuclear energy systems requires a deep understanding of atomic behavior.
During nuclear fission and fusion reactions, atoms often transform into other atoms with the same mass number but different atomic numbers. Studying these transformations allows scientists to monitor nuclear reactions, manage radioactive waste, and improve reactor efficiency.
Research in this area helps create safer nuclear energy technologies while reducing environmental risks.
Conclusion
Nuclear chemistry is a complex yet essential scientific field that helps us understand the behavior of atomic nuclei. One of its key concepts involves atoms that share the same mass number but differ in their atomic structure. These atoms play a critical role in studying nuclear reactions, radiometric dating, medical imaging, environmental science, and nuclear energy research.
By examining Isobaric Compounds, scientists gain deeper insights into how atoms transform and interact during nuclear processes. This knowledge supports advancements in technology, medicine, and environmental protection.
Organizations such as Iroa Technologies continue to support scientific progress by providing analytical tools that help researchers explore complex chemical and nuclear data. As nuclear science continues to evolve, the study of these atomic structures will remain essential for future discoveries.
FAQs
1. What are isobaric compounds in nuclear chemistry?
Isobaric compounds are atoms that share the same mass number but have different atomic numbers. This means they contain different numbers of protons and neutrons.
2. Why are these compounds important in nuclear research?
They help scientists study nuclear reactions, radioactive decay, and atomic stability, which are essential for understanding nuclear processes.
3. How are they used in radiometric dating?
They help track the transformation of radioactive atoms into other elements with the same mass number, allowing scientists to estimate the age of geological or archaeological samples.
4. What role do they play in nuclear medicine?
They are used in the development of radioactive tracers and imaging techniques that help doctors diagnose diseases and monitor organ function.
5. How do analytical technologies help study these compounds?
Advanced laboratory tools, including those supported by companies like Iroa Technologies, help scientists detect and analyze atoms with identical mass numbers but different atomic structures.